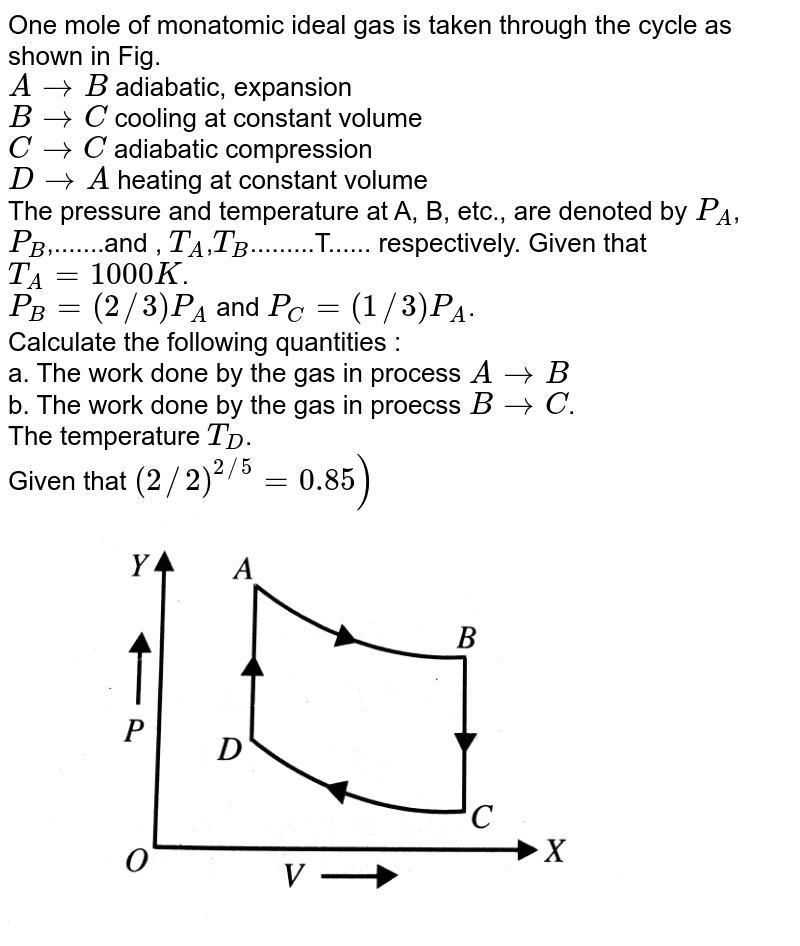
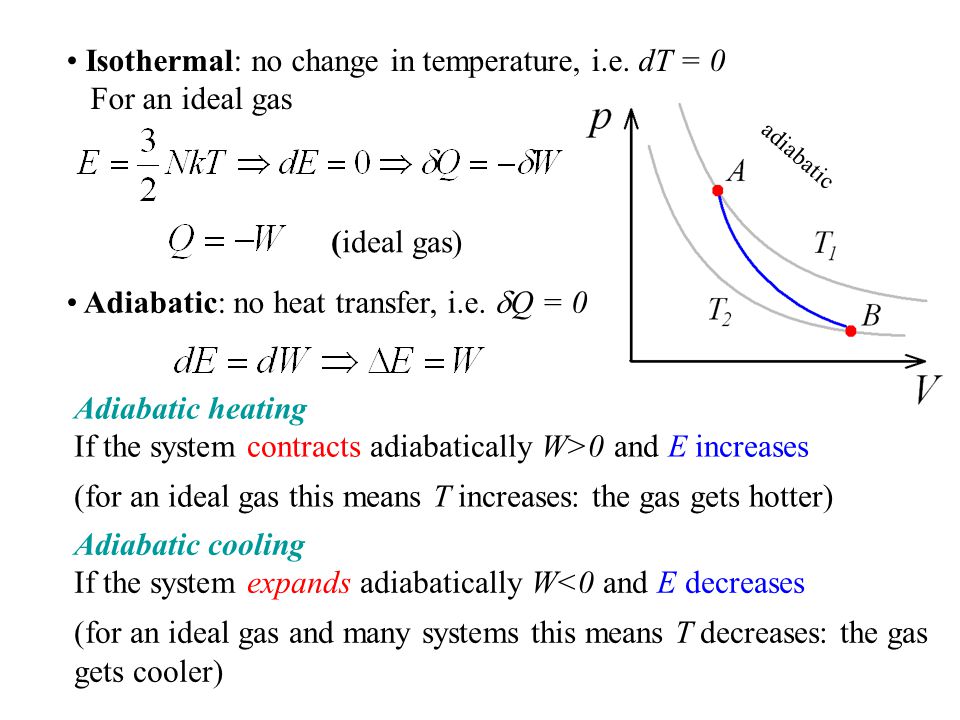
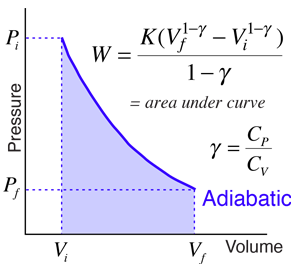
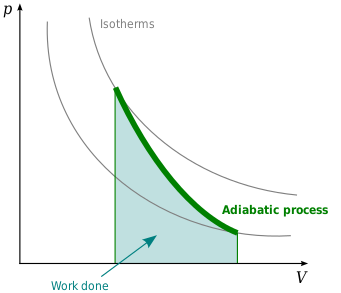
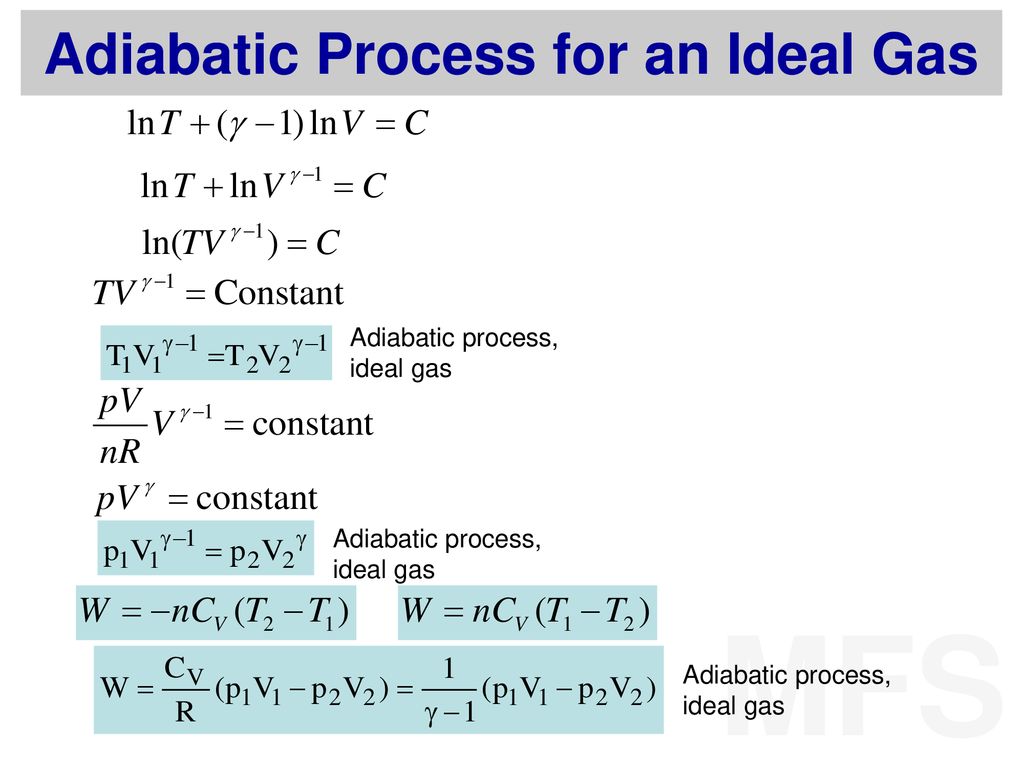
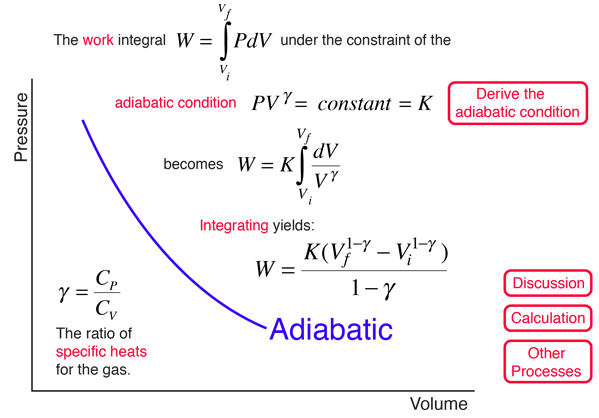
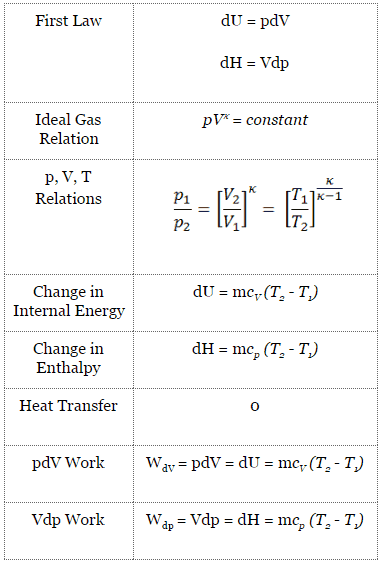One mole of a monatomic ideal gas is taken through the cycle shown in the figure: A to B adiabatic expansion <img src="https://d10lpgp6xz60nq.cloudfront.net/physics_images/VMC_PHY_XI_WOR_BOK_03_C10_E05_076_Q01.png" width="80%"> B to C: cooling at ...
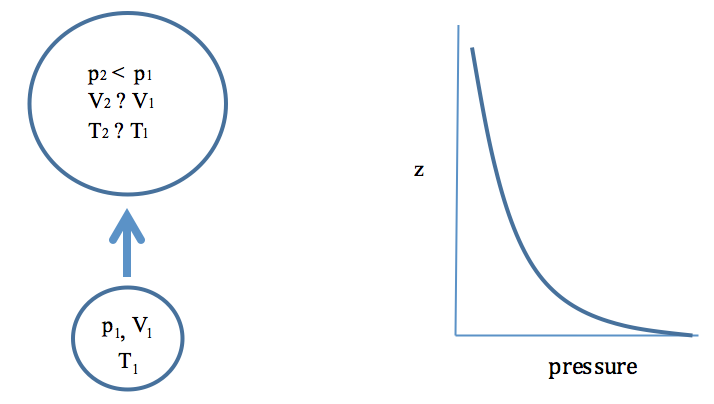
2.5 Adiabatic Processes: The Path of Least Resistance | METEO 300: Fundamentals of Atmospheric Science