Märklin H0 83307 Delta Digital Dampflok T 18, - Spielzeug 09.11.2020 - Erzielter Preis: EUR 95 - Dorotheum

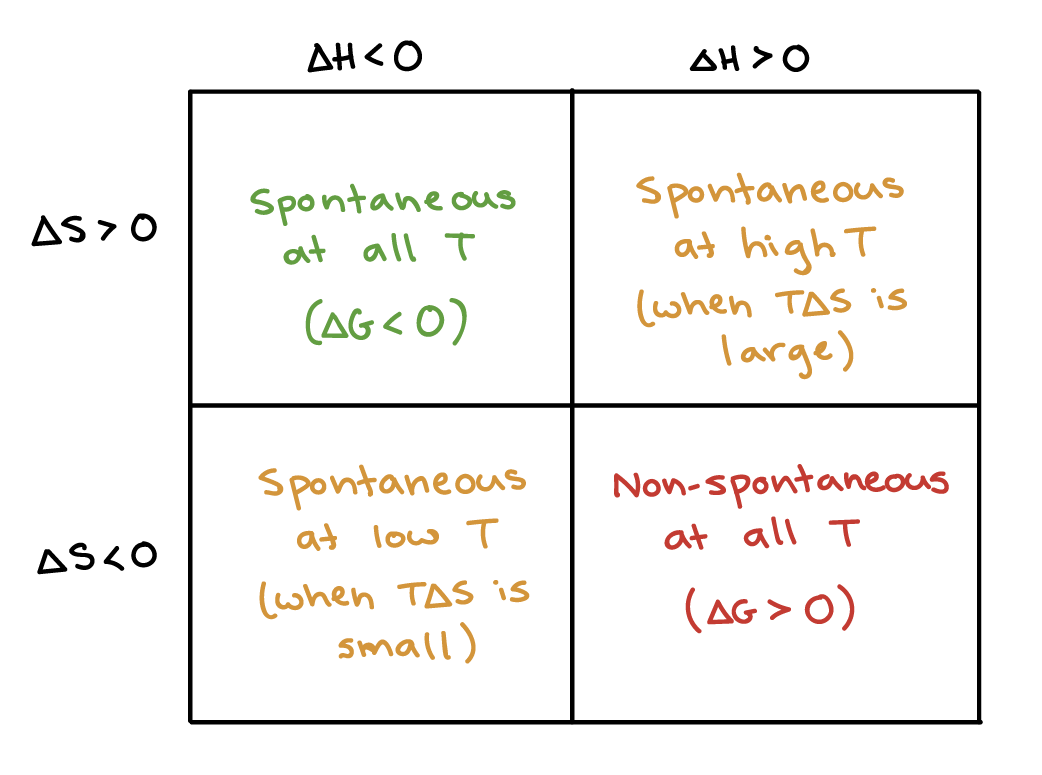
1 H = H final - H initial If H final > H initial then H is positive Process is ENDOTHERMIC If H final > H initial then H is positive Process is ENDOTHERMIC. - ppt download

A particular reaction at 27^(@)C for which DeltaH gt 0 and DeltaS gt 0 is found to be non-spontaneous. The reaction may proceed spontaneously if

SOLVED: Which of the following is true for any system in a state of equilibrium? 0 Q<0 0 Delta H > 0 0 k>1 0Q-k

OneClass: Of the following, Delta H degree f is not zero for ______. A) O_2 (g) B) C (diamond) C) ...



![Solved Calculate [delta H0 f] in kJ for the equation below | Chegg.com Solved Calculate [delta H0 f] in kJ for the equation below | Chegg.com](https://media.cheggcdn.com/media/b5e/s700x491/b5e608eb-b559-4cd4-8e51-3484cc1169cf/image.png)

![filters - Causal inverse of $h[n]=\delta[n]-\alpha\delta[n-1]$ - Signal Processing Stack Exchange filters - Causal inverse of $h[n]=\delta[n]-\alpha\delta[n-1]$ - Signal Processing Stack Exchange](https://i.stack.imgur.com/yJWqp.jpg)







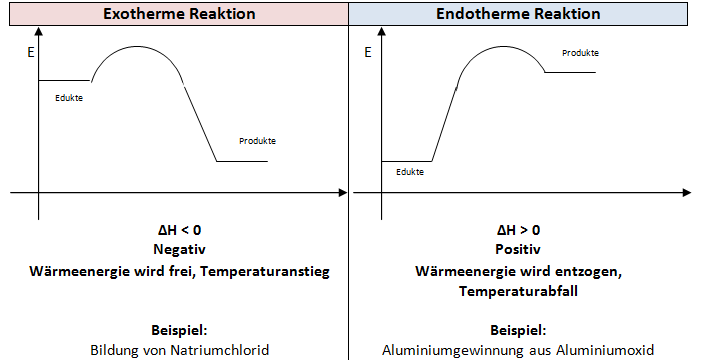
![Enthalpie Definition und Formel verständlich erklärt · [mit Video] Enthalpie Definition und Formel verständlich erklärt · [mit Video]](https://d1g9li960vagp7.cloudfront.net/wp-content/uploads/2019/03/Enthalpie-und-Reaktionsenthalpie_SEO-1024x576.jpg)