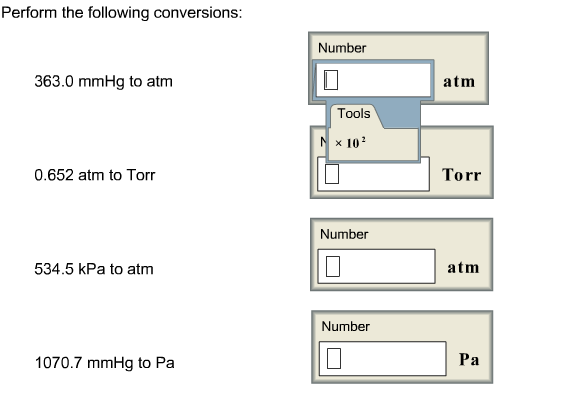
Warm up 5-2-16 1.Convert 65.0 mmHg to atm Reference: 1 atm = 760 mmHg = 101.325 kPa 2. A helium balloon has a volume of 2.75 L at 20 ºC. the volume decreases. - ppt download
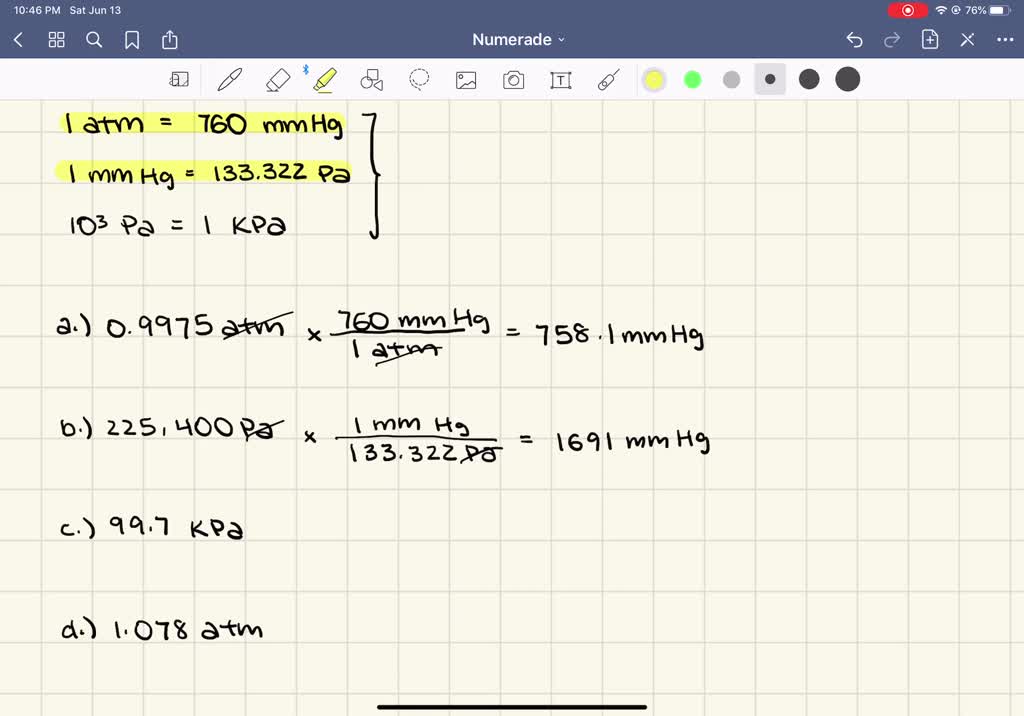
SOLVED:Convert the following pressures into units of mm Hg. a. 0.9975 atm b. 225,400 Pa c. 99.7 kPa d. 1.078 atm