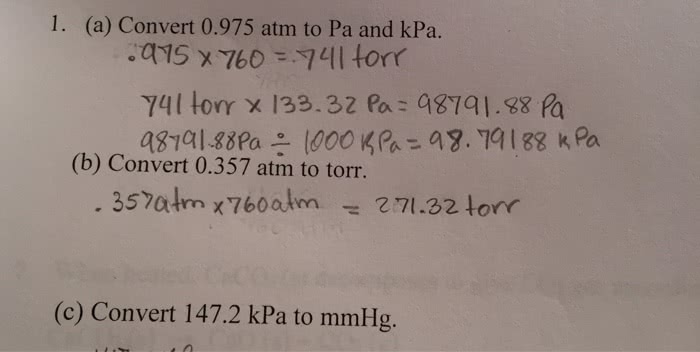
OneClass: 1. (a) Convert 0.975 atm to Pa and kPa. .qy5 x 760 = 41 torr (b) Convert 0.357 atm to torr....

SOLVED:Make the indicated pressure conversions. a. 1.54 \times 10^{5} Pa to atmospheres b. 1.21 atm to pascals c. 97,345 Pa to mm Hg d. 1.32 kPa to pascals

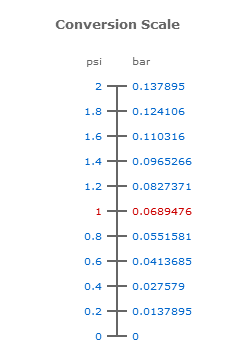
Atmospheres and Conversions: 1.00 atm = x 10 5 Pa = kPa = 760. Torr = 14.7 psi To convert pressure: 1.Turn what you have to atm 2.Multiply. - ppt download
![13-2: Measuring Gases. Variables There are four different variables to consider when measuring gases: 1.Amount of Gas: [n] usually measured in moles. - ppt download 13-2: Measuring Gases. Variables There are four different variables to consider when measuring gases: 1.Amount of Gas: [n] usually measured in moles. - ppt download](https://images.slideplayer.com/27/9178436/slides/slide_6.jpg)
13-2: Measuring Gases. Variables There are four different variables to consider when measuring gases: 1.Amount of Gas: [n] usually measured in moles. - ppt download











/sun-beams-diffuse-ocean-micronesia-palau-128941208-587b91ad5f9b584db36312c9.jpg)